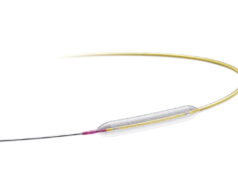
Spectranetics has been granted the CE mark for its AngioSculptX Drug-coated percutaneous coronary transluminal angioplasty (PCTA) scoring balloon catheter. A press release reports that the balloon is a “first-of-its-kind” device as it combinines the proven AngioSculpt PTCA scoring balloon catheter with a drug coating. The device is indicated for the treatment of haemodynamically significant coronary artery stenosis, including in-stent restenosis.
Bruno Scheller (Saarlandes University Clinic in Homburg, Germany) comments: “AngiosculptX represents a unique coronary scoring technology that incorporates a drug coating. Given its clinical results, it is a significant advancement in therapy options for patients.”
As previously disclosed, and part of the original acquisition of AngioScore, there is a US$5million milestone payment related to receiving CE mark for AngioSculptX that the Spectranetics expects to pay in the third quarter.