 Boston Scientific has signed an agreement to acquire Claret Medical, which developed and commercialised the Sentinel cerebral embolic protection system. The device is used to protect the brain during certain interventional procedures, predominately in patients undergoing transcatheter aortic valve implantation (TAVI). The transaction price consists of US$220 million in up-front cash, as well as a potential reimbursement-based milestone payment of up to US$50 million.
Boston Scientific has signed an agreement to acquire Claret Medical, which developed and commercialised the Sentinel cerebral embolic protection system. The device is used to protect the brain during certain interventional procedures, predominately in patients undergoing transcatheter aortic valve implantation (TAVI). The transaction price consists of US$220 million in up-front cash, as well as a potential reimbursement-based milestone payment of up to US$50 million.
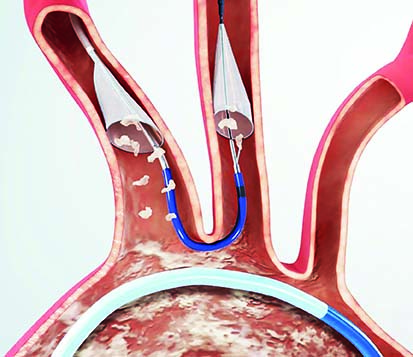
The Sentinel system—which received CE mark in 2014 and US FDA clearance in 2017—is the only device cleared to protect patients against the risk of stroke during TAVI (in both the US and the EU). A press release reports that recent studies have estimated approximately 4% of patients experience a clinically-apparent stroke within 30 days of a TAVI procedure.
In the SENTINEL trial, which led to FDA clearance and commercial introduction of this therapy in the USA, the Sentinel system reduced the incidence of strokes by 63% within the first 72 hours of the procedure. In clinical studies, the system captured debris flowing towards the brain in 99% of TAVI cases—regardless of the type of replacement valve that was used.
Kevin Ballinger, president, Interventional Cardiology, Boston Scientific, comments: “Through the development and commercialisation of the Sentinel System, Claret Medical has successfully introduced a new layer of safety and peace of mind for physicians and their patients undergoing TAVI procedures. This acquisition will expand our commercial portfolio to include an important adjunctive offering aimed at improving TAVI patient outcomes. We also see potential for future use in other left heart and endovascular procedures such as mitral valve repair and replacement, left atrial appendage closure and pulmonary vein isolation ablation procedures for atrial fibrillation.”
Claret Medical is based in Santa Rosa, USA, and has approximately 120 employees. The acquisition is projected to close during the third quarter of 2018, subject to customary closing conditions.
The transaction is expected to be immaterial to adjusted earnings per share in 2018, accretive in 2019 and increasingly accretive thereafter. On a GAAP basis, the transaction is expected to be less accretive, or more dilutive as the case may be, due to amortization expense and acquisition-related net charges.
Azin Parhizgar, president and chief executive officer, Claret Medical, says: “We have been pleased by the consistent, high-quality clinical evidence generated by the committed thought leaders at leading academic centres, as well as the rapid physician adoption of the device—used on average in 60% of TAVI procedures performed across 100 centres—within just 12 months of carefully-controlled US commercialisation. Nearly 10,000 patients have been treated worldwide with the Sentinel System, and we are confident that the leadership of Boston Scientific will enable increased momentum and improved patient access to this valuable technology.”









