Abbott has announced that the US Food and Drug Administration (FDA) has approved the company’s CardioMEMS HERO device—a next-generation pulmonary artery (PA) pressure reader—to support the care of people living with heart failure.
The reader contains new design features to help empower heart failure patients and their clinicians with the daily insights they need to detect and combat heart failure progression, the company says in a press release. Abbott will soon begin commercial release of the CardioMEMS HERO reader in the USA.
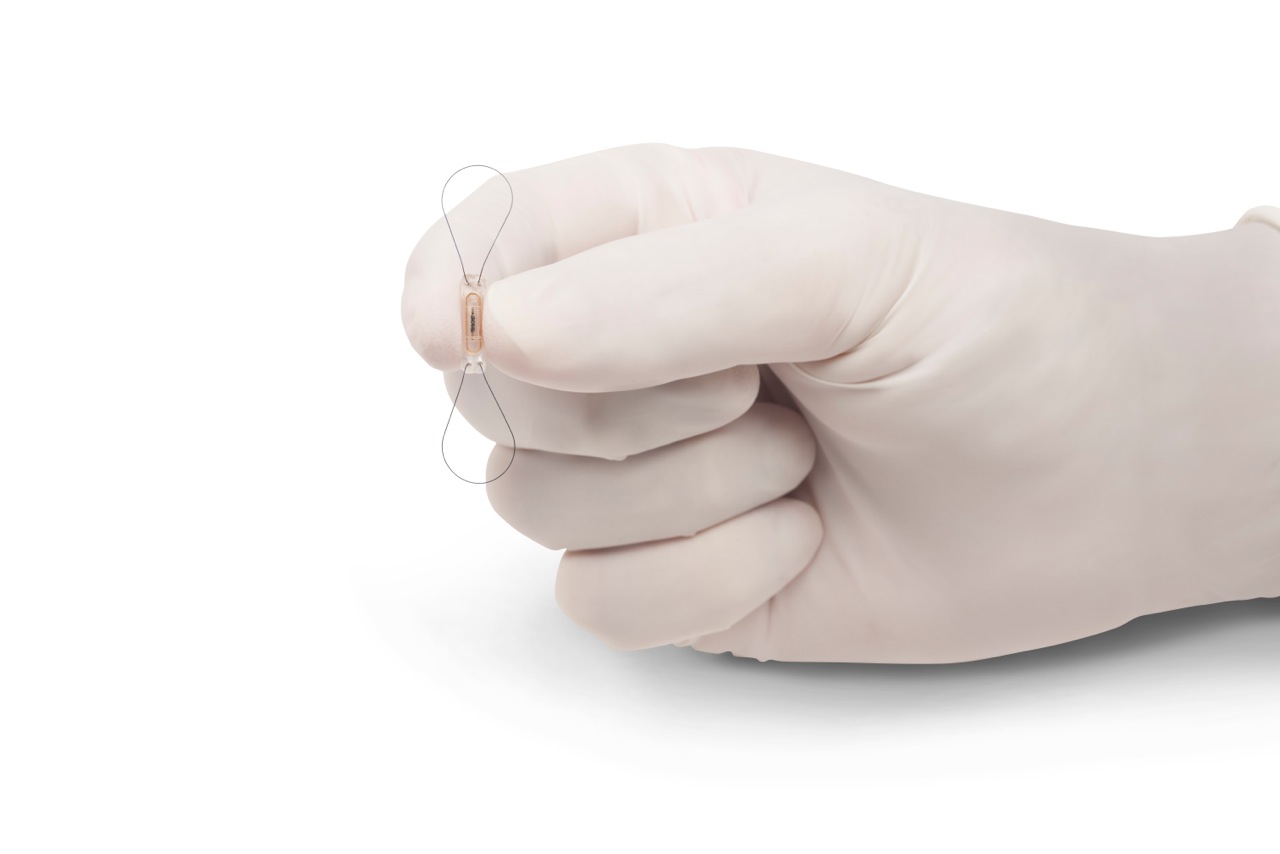
Abbott’s CardioMEMS HERO reader is a new component of the CardioMEMS HF system. The HERO reader is similar in size to a laptop case and is sleeker and smaller than the system’s previous model. It is designed to work with the CardioMEMS PA sensor—a paperclip-sized device implanted near the heart where rising PA pressures signal worsening heart failure.
Patients lay with their back centered on the HERO reader for less than 60 seconds on average to take a remote PA pressure reading and securely send the data to their care team. Clinicians will receive the daily data readings and if they see changes in PA pressures, they will communicate to the patient any medication or lifestyle changes needed to help slow heart failure progression.
“Data from CardioMEMS HF system clinical trials show the positive impact the device has on reducing the risk of heart failure hospitalisations and cardiovascular death. HERO is likely to significantly enhance data acquisition and patient interactions with this life-changing technology,” said JoAnn Lindenfeld, director of advanced heart failure at Vanderbilt University Medical Center (Nashville, USA), who served as primary investigator for the GUIDE-HF study. “HERO is lighter, more comfortable, and easier to transport and use which improves patient experience.”